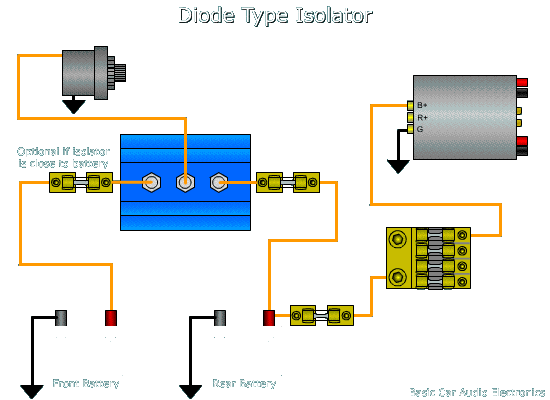 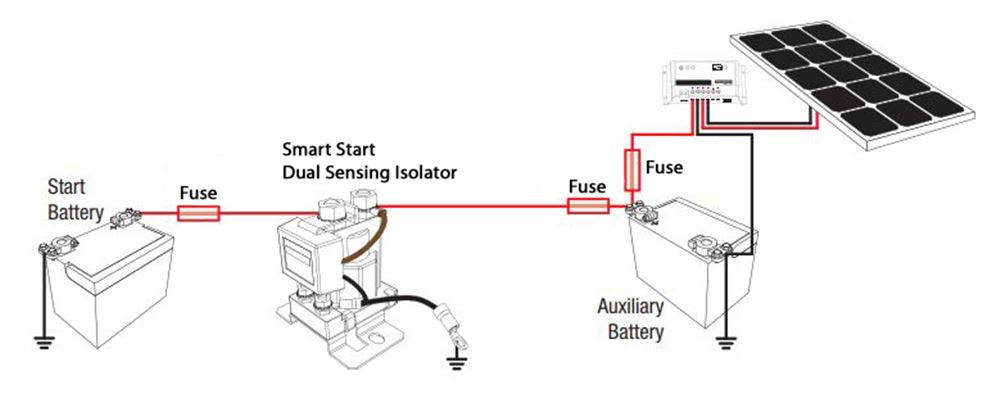
The electrolyte provides a means for ion migration whereby ions move to prevent charge build-up that would otherwise stop the reaction. The presence of an electrolyte and an electrical conducting path between the metals is essential for galvanic corrosion to occur. This leads to the metal at the anode corroding more quickly than it otherwise would and corrosion at the cathode being inhibited. 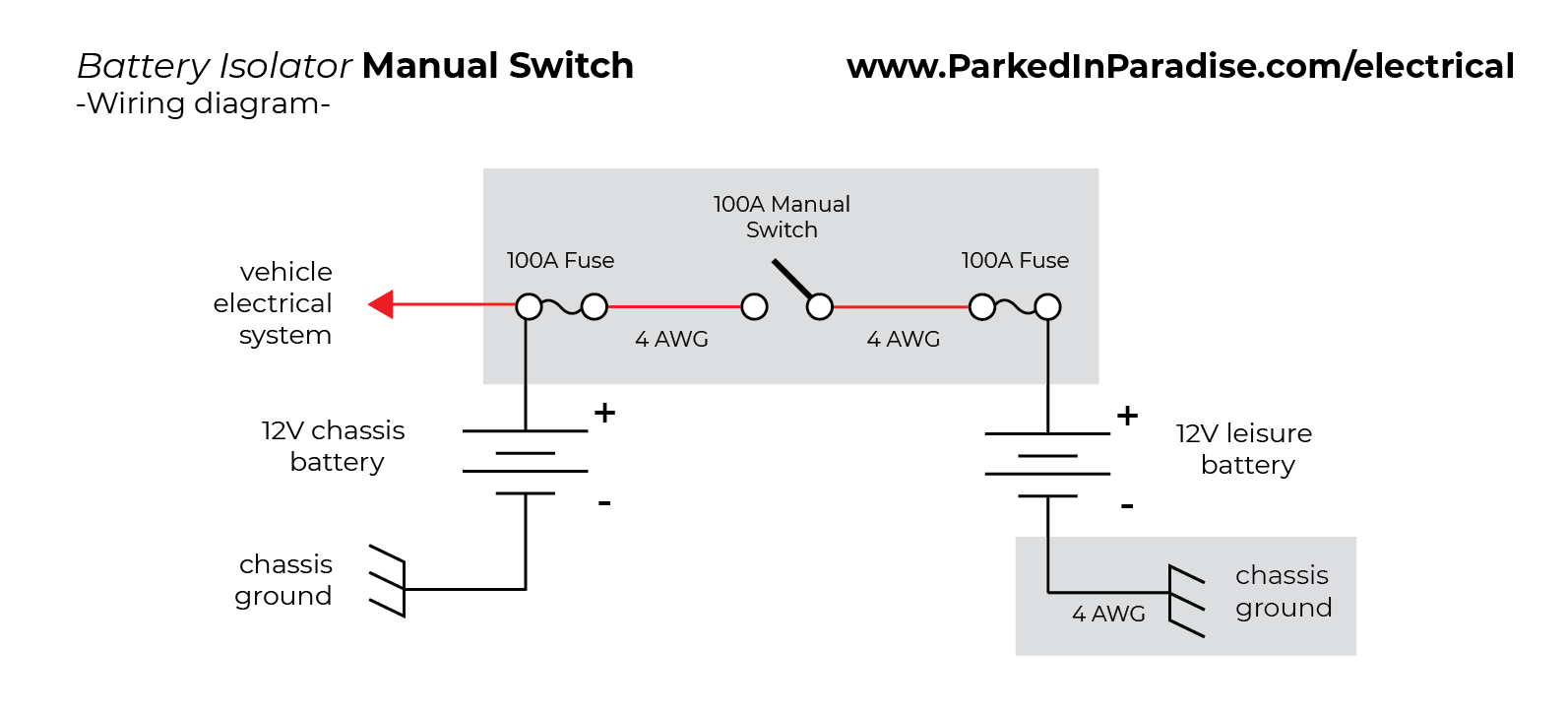
The electropotential difference between the reactions at the two electrodes is the driving force for an accelerated attack on the anode metal, which dissolves into the electrolyte. This phenomenon is named after Italian physician Luigi Galvani (1737-1798).ĭissimilar metals and alloys have different electrode potentials, and when two or more come into contact in an electrolyte, one metal (that is more reactive) acts as anode and the other (that is less reactive) as cathode. A similar galvanic reaction is exploited in primary cells to generate a useful electrical voltage to power portable devices. Galvanic corrosion (also called bimetallic corrosion or dissimilar metal corrosion) is an electrochemical process in which one metal corrodes preferentially when it is in electrical contact with another, in the presence of an electrolyte. Corrosion of an iron nail wrapped in bright copper wire, showing cathodic protection of copper a ferroxyl indicator solution shows colored chemical indications of two types of ions diffusing through a moist agar medium
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
